Unraveling the 'Persistence of Totipotency': How Mechanical Forces Shape Embryonic Development and Cancer
In a groundbreaking research paper by Jaime Cofre, titled "Mechanical cues for totipotency and the preneural state: embryo and cancer expanding the frontiers of developmental physics," the author delves into the remarkable role that physics plays in the intricate processes of cell differentiation and embryonic development, particularly in cancer biology. The paper presents a compelling argument that mechanical forces, rather than merely chemical signals, are fundamental in guiding the creation and alignment of biological structures, influencing everything from embryonic stem cell behavior to the onset of cancerous growths.
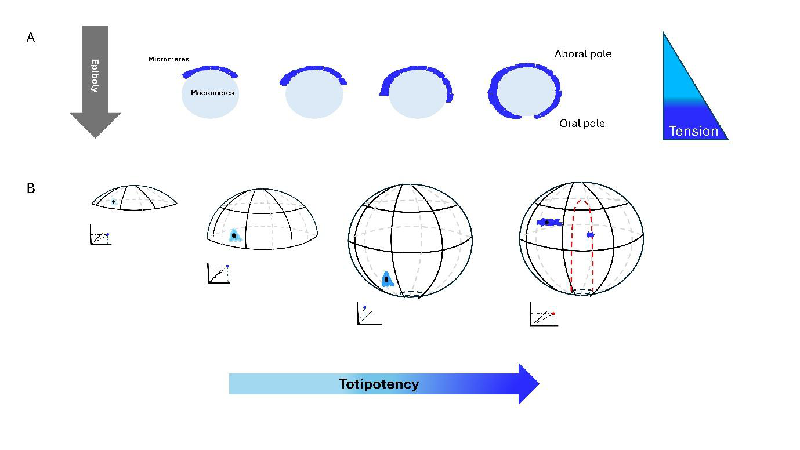
Understanding Totipotency through Mechanical Memory
Central to Cofre's argument is the concept of 'totipotency,' the remarkable ability of a zygote to develop into any cell type in an organism. He posits that this capacity is not solely rooted in genetic makeup but is also encoded in the physical and mechanical history of cells, which he refers to as 'persistent mechanical memory.'
When embryonic cells undergo processes such as 'epiboly' (the expansion of cell layers), they create and react to mechanical forces that shape their development. As these forces are exerted, they leave a lasting imprint on the cells, which assists in maintaining the totipotent state throughout development. This perspective shifts the traditional focus from a purely genetic understanding of development to a more integrated view that includes physical forces as major players.
The Connection between Embryology and Cancer
One of the most fascinating aspects of Cofre’s research is the parallel he draws between embryonic development and cancer. He identifies that both processes share a common thread in cell differentiation, with each exhibiting responses to mechanical forces. Just as cells undergo differentiation during normal development, cancer cells, when influenced by their mechanical environment, can also exhibit similar transformative behaviors.
By understanding the physics underlying these processes, researchers can uncover potential therapeutic strategies for manipulating cell behavior in diseases like cancer, where aberrant growth signals can be mitigated by mechanical intervention.
Implications for Future Research and Medicine
This innovative merger of mechanics and biology opens new pathways for research into regenerative medicine and cancer therapy. As mechanobiology emerges as a vital field, the insights drawn from embryonic studies can enhance methods for tissue engineering and cellular therapies, potentially improving outcomes in treating diseases characterized by abnormal cell behavior.
To appreciate the full spectrum of developmental biology, it becomes imperative to explore how physical principles can be applied to our understanding of life at its most fundamental level. The shift from viewing biological phenomena through a strictly chemical lens towards a more holistic view that incorporates physics represents a seismic change in how we approach life sciences.
In conclusion, this paper not only challenges existing paradigms in embryology and cancer research but also encourages the scientific community to actively integrate mechanical considerations into developmental biology—a move that could fundamentally reshape our understanding of how life evolves and how we may treat life-threatening diseases.