Unlocking the Secrets of Brain Diffusion: How Geometry Shapes Molecular Movement
Recent research conducted by a team of scientists from renowned institutions has shed new light on the intricate dynamics of molecular motion in the brain's extracellular space (ECS). Their study, titled "fScale-Dependent Emergence of Hindered Diffusion in the Brain Extracellular Space," reveals that the slowing down of diffusion in brain tissues is not merely due to a single property but arises from the complex geometry of the ECS itself.
Understanding the Brain's Extracellular Space
The ECS is a densely packed environment in the brain that plays a crucial role in supporting neuronal function and communication. Traditional descriptions of molecular movement within this space often relied on the concept of "tortuosity," which describes how diffusion is hindered compared to a free solution. However, this study suggests that tortuosity is, in fact, an emergent property influenced by various factors, rather than a fixed parameter.
A Novel Methodology for Visualizing Diffusion
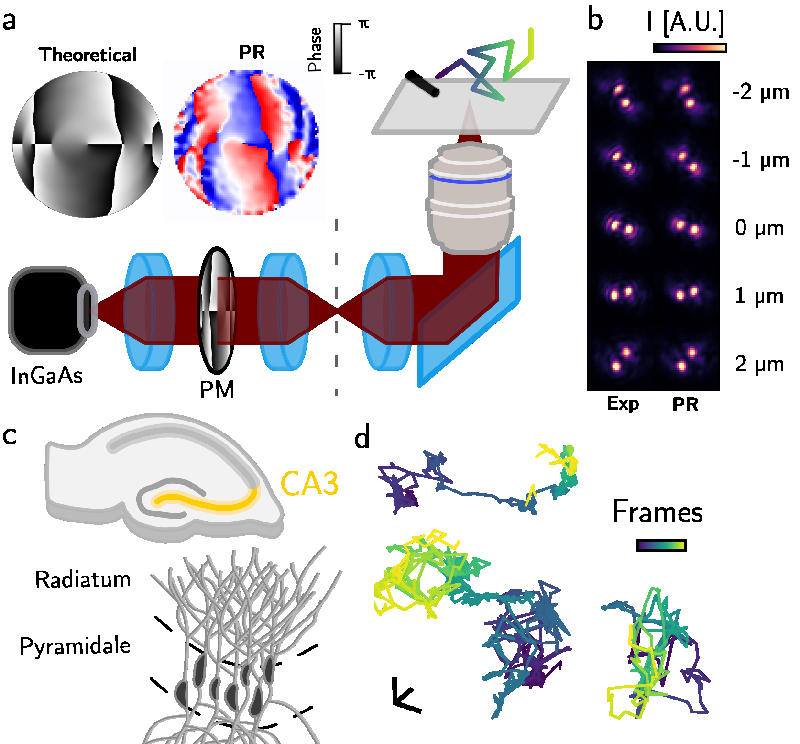
The researchers employed cutting-edge techniques involving ultrashort carbon nanotubes (uCCNTs) as tracers, allowing them to visualize molecular motion in three dimensions with remarkable precision. By doing so, they could observe how molecules behave at different scales and how their motion becomes constrained as they navigate through the complex architecture of brain tissue.
This innovative approach enabled the scientists to identify two distinct regimes of molecular transport: at shorter length scales, molecules exhibited what can be described as 'free' diffusion, while at longer scales, the motion transitioned to a more restricted form—reflecting the constraints imposed by the structural layout of the ECS.
Key Findings and Implications
The study unveiled that the effective diffusivity of molecules in the ECS can be reduced by factors of 2 to 5 compared to diffusion in free solution. This reduced mobility can have significant implications for brain function, as it may affect how signaling molecules and nutrients are distributed among neurons. Essentially, while molecules can move freely over short distances, their ability to traverse larger scales is hindered by the surrounding cellular structures.
Furthermore, the research highlights that the unique architecture of the ECS could influence the behavior of various biomolecules such as neurotransmitters and could play a pivotal role in processes like learning and memory. The interplay between molecular movement and the complex geometry of brain tissue may thus be a critical factor in understanding various neurological conditions and cognitive functions.
Looking Ahead
As this research opens up new questions about how molecular diffusion operates within living tissues, it sets the stage for further exploration into the role of geometry in biological systems. By continuing to investigate these dynamics, scientists hope to unravel more intricacies of brain physiology and develop targeted strategies for addressing disorders associated with disrupted molecular transport.
Overall, the insights gained from this study represent a significant advancement in our understanding of brain function, bridging the gap between biology and physics while emphasizing the remarkable complexity of our brains' architecture.
Authors: Quentin Gresil, Ivo Calaresu, Finn L. Sebastian, Benjamin Flavel, Jana Zaumseil, Laurent Groc, Laurent Cognet